Rationale for Limit Selection in Process Validation
Recommendation

Tuesday, 5 May 2026 9 .00 - 16.15 h
In a recent warning letter, the US FDA criticised deficiencies in the process validation of a tablet manufacturing process. In particular, the FDA questioned the scientific justification for the selected process parameter limits.
The warning letter refers to the setting of the compression force limits, which were defined with upper and lower limit, commonly used. In the case of the Chinese manufacturer, rejection limits were chosen based on previous manufacturing experience. This is precisely what the FDA criticised in terms of a lack of process validation. For at least one batch, a deviation related to dissolution specifications was reported, which was attributed to excessive compression force.
The FDA reiterated its expectations regarding process validation and referred to its process validation guidance.
The company's response following the FDA inspection included preparing additional batch documentation and defining compression force limits based on rejection studies. However, the FDA considered this response insufficient.
Why?
The FDA criticised that compression force limits were justified mainly in relation to tablet weight. From the FDA's perspective, this rationale was incomplete, as other critical quality attributes - such as tablet hardness - were neither adequately evaluated nor incorporated into the control strategy. The FDA therefore requires:
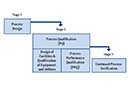
- A comprehensive summary of the validation programme demonstrating a continuous state of control throughout the product lifecycle, including associated procedures
- A detailed description of the process performance qualification (PPQ) programme and ongoing monitoring of intra- and inter-batch variability
- A timeline for performing PPQ activities for all marketed products
- PPQ plans and written procedures for qualification of equipment and facilities
- A comprehensive programme covering process design, validation, maintenance, control and monitoring of all manufacturing processes, including careful monitoring of variations within and between batches to ensure a continuous state of control. Besides, a programme for the qualification of equipment and facilities.
Furthermore, the FDA recommended engagement of an independent GMP consultant to address broader compliance deficiencies.
Conclusion: Rationales play an important role in the GMP environment. The response from the inspected company to the FDA regarding the inadequate process validation clearly illustrates the importance of this.
The full warning letter con be found on the FDA website.