Risk Assessment as the Basis for Process Validation
Recommendation

Tuesday, 5 May 2026 9 .00 - 16.15 h
In a recent Warning Letter, the FDA criticised the fact that not all parameters of a process were included in a process validation. What are the follow-up measures?
Referring to 21 CFR 211.100 (a), the FDA stated: "Your firm failed to establish adequate written procedures for production and process control designed to assure that the drug products you manufacture have the identity, strength, quality, and purity they purport or are represented to possess."
In general, the FDA criticised the insufficient validation of the manufacturing process - as well as the missing parameters in procedures and in batch documentation that are relevant to quality. The authority further addressed the lack of qualification of production equipment. So much for the deficiencies.
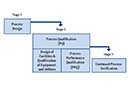
With reference to the FDA process validation guideline, the FDA outlined what it understands by process validation. In the Warning Letter, it described the validation life cycle, beginning with the development of a product and the implementation of the control strategy. Process qualification studies are then to demonstrate that the control strategy works.
What does the FDA require?
Following the FDA directly required:
- A risk assessment and the follow-up measures necessary for products on the market without process validation.
- A detailed risk assessment addressing the potential problems of the systems under consideration with regard to medicinal products on the market within the expiry date.
- Measures such as customer information and product recalls based on the above risk assessment.
Repeated inspection findings
The FDA further pointed out in the Warning Letter that the same deficiencies were found in a previous inspection in 2019. Although the company promised to take corrective action, nothing happened. The FDA considered this to be a failure on the part of senior management, which should have an overview of the manufacturing process.
CGMP consultant recommended
Due to the nature of the inspection findings, the FDA recommended to consult a CGMP consultant. Their qualifications should meet the requirements of 21 CFR 211.34. However, calling in a consultant does not exempt senior management from their responsibility to remedy the deficiencies and comply with CGMP.
Conclusion: Process validation covers all process steps and all critical parameters. Indirectly, the FDA also requires a risk assessment to identify these. Furthermore, products that are on the market without validation must be critically examined.
Please see the FDA website for the entire Warning Letter.