What does the Concept Paper on Annex 15 Revision say? A detailed Analysis
In the previous newsletter, you could read about a concept paper from the EMA describing the planned changes to Annex 15 (Qualification and Validation) of the EU GMP Guidelines. A detailed analysis is now provided below.
In response to the nitrosamine contamination in sartans, one of the causes identified was insufficient process and product knowledge during development. This prompted the EMA to undertake a further revision of Annex 15 on validation/qualification. The main objective of the revision is to make the inclusion of active substances in Annex 15 mandatory. Until now, it has only been optional for active substance manufacturers. This affects both manufacturers of chemical active substances and biological active substances. The changes in quality risk management included in the revision of ICH Q9 (R1) are also to be incorporated.
What exactly is planned?
The Discussion section provides further details, which also primarily concern active substances.
- The concept of a validation master plan (referred to as a 'validation master file' in the concept paper), the concept of a validation policy and the concept of change control are to be extended to active substances. This is expected to lead to improvements among active substance manufacturers with regard to the definition and documentation of qualification and validation activities. This applies in particular to the creation of validation protocols, as described in Part II of the EU GMP guidelines for active substances. Change control is regarded as an important element of knowledge management.
- More expectations regarding validation by third-party providers are to be included so that active substance manufacturers gain more control over outsourced activities.
- The need for investigations in the event of deviations from the predefined acceptance criteria is also to be expanded. The hope here is that these expansions will encourage active substance manufacturers to develop a deeper understanding of the process.
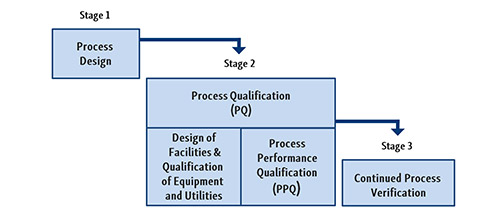
- In addition to the qualification levels DQ, IQ, OQ and PQ already known in the active substance sector, user requirement specifications (URS), factory acceptance tests (FAT) and site acceptance tests (SAT) are now also to be introduced by active substance manufacturers. These measures are also expected to strengthen control over the processes.
- The changes are also intended to reinforce more robust process development and clarify expectations for concurrent validation. Expectations for supplier qualification are also to be emphasised more strongly. Particular attention will be paid to process validation in the context of the reuse of materials and solvents. When creating a validation protocol, focus will also be placed on variables that have an influence on critical quality attributes. Assistance with continuous process verification and the 'hybrid' approach is also to be expected. Likewise, greater emphasis will be placed on periodic reviews.
- Similar to the verification of transport for medicinal products, this topic will also be included in relation to active substances, taking into account product knowledge and the influence on quality during transport.
- With regard to monitoring systems, greater emphasis will be placed on quality risk management (QRM) in development and qualification/validation in the revision. There are also plans to provide assistance with QRM with regard to risk reviews in the context of validation and qualification activities. This is also intended to establish a link to Chapter 2 of the EU GMP Guidelines Part II.
- In future, there should also be a greater emphasis on QRM in traditional process validation.
Further possible amendments to Annex 15
The concept paper further describes the possibility that the Inspectors Working Group and the PIC/S Sub-committee on GMDP Harmonisation, which is also involved, may also take into account other relevant points or changes that arise during the development of the revision.
Comments on the concept paper can be submitted until 9th April 2026. A first draft document on the revision is planned for the end of 2026. The ECA Validation Group will comment the concept paper.
Conclusion: The main focus of the planned second revision of Annex 15 is on the full integration of active substances. Specifications on accompanying validation and quality risk management could also be relevant for drug manufacturers.